 PF 3Cl 2 - Phosphorus Trifluoride DichlorideĤ4. SF 5Cl - Sulfur Pentafluoride MonochlorideĤ1. SF 5Cl - Sulfur Monochloride PentafluorideĤ0. The Valence Shell Electron Pair Repulsion (VSEPR) theory is a simple and useful way to predict and rationalize the shapes of molecules. Step 3: Use symmetry to determine if the molecule is polar or non-polar.Ĭlick on the molecule's name to see the answer, but first try to do it yourself!ġ. Step 1: Draw the Lewis structure, Step 2: Draw the 3D molecular structure w/ VSEPR rules, 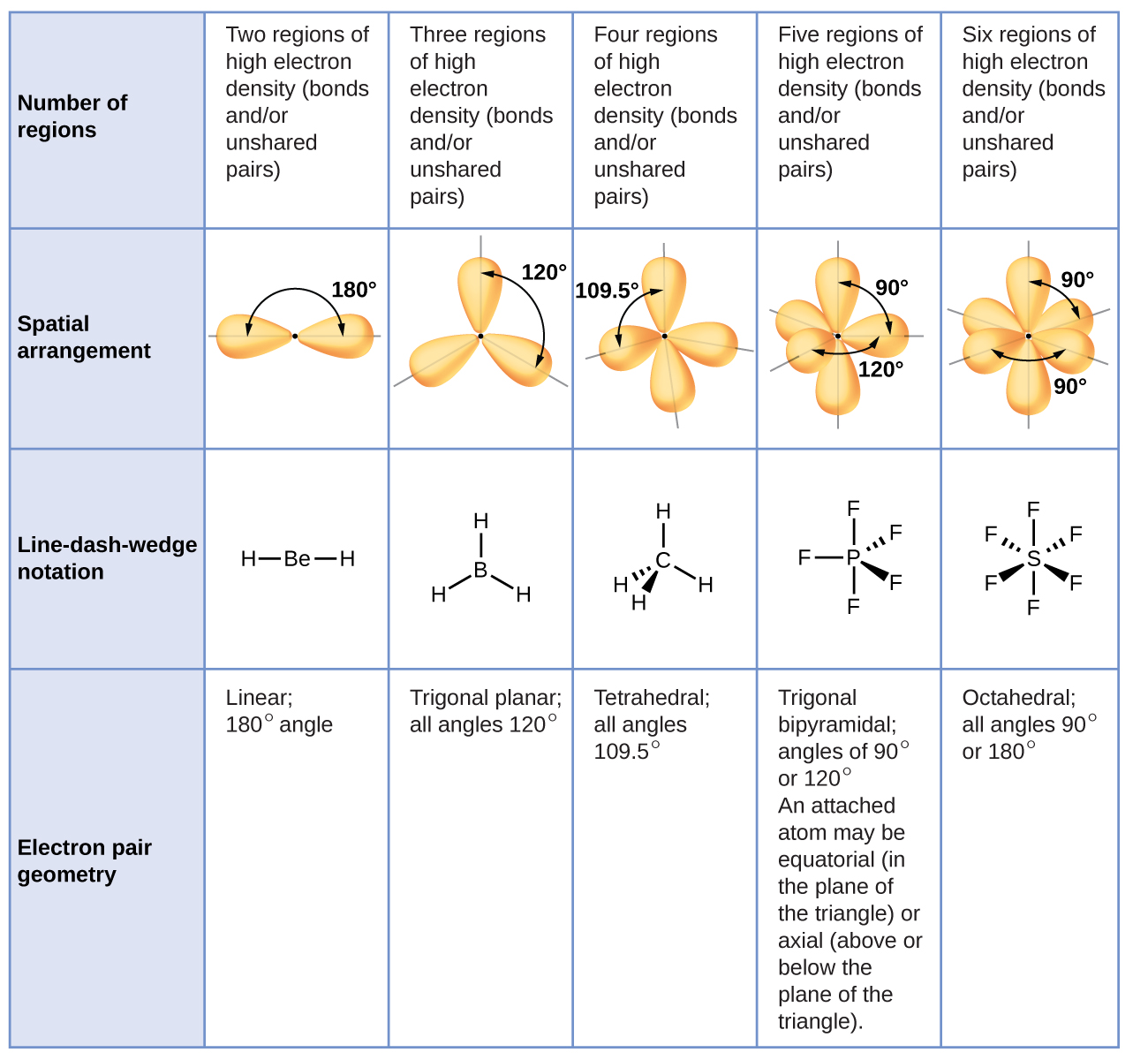
Molecular Geometry & Polarity Example Problems VSEPR Rules: Electron and Molecular Geometry On Central Atom. VSEPR Rules: Table of Molecular Geometry, Molecular Polarity Problems (with 3D solutions!). And it's a good thing, because if water was not so polar, we would certainly not be here. Water, for example, is a very light molecule (lighter than oxygen gas or nitrogen gas) and you might expect it would be a gas based on its molecular weight, however the polarity of water makes the molecules "stick together" very well. when you compare it to other similar molecules. The polarity of a molecule will tell you a lot about its solubility, boiling point, etc. If the atoms are the same, the molecule is non-polar molecule if the atoms are different, the molecule is polar. Note: molecules with two atoms are not shown in these examples they are always linear with sp hybridization. If you click on the example molecules (where it says 3D view) below you'll get a better understanding of why some molecules are polar and some not. You need to consider the molecule in 3D (three dimensions). To really understand how to do this, the Lewis structure is only the first step. However, if the molecule is asymmetric, the bond dipole moments won't "cancel out" and the molecule will have a net dipole moment and the molecule is therefore polar. Step 3: Determine if the molecular is polar or non-polar - a molecule is (i) non-polar if the charge distribution is symmetric and (ii) polar if the charge distribution is asymmetric (not symmetric).Īfter you draw the molecule in 3D representation using VSEPR rules, if the molecule has symmetry around the central atom, the bond dipole moments will "cancel out" (like pulling in opposite directions) and the molecule will therefore be non-polar. 
According to the VSEPR theory, the two sets of electrons will orient themselves 180. Note that double bonds and triple bonds count as a single region of electrons. This compound has two groups of electrons around the carbon. Valence Shell Electron Pair Repulsion theory around the central atom all regions of electrons repel each other to get as far away from each other as possible while pivoting around the central atom. The statement VSEPR model is used to determine bond polarity is not true because the VSEPR model is usually used to identify the compounds molecular. Note the number of electron regions around the central atom, and of these which are bonding or lone pairs (non-bonding pairs) Step 2: Use this info to determine the 3D geometry of the molecule. These are problems using 3D molecules run in the application Jmol to help you visualize the molecule to determine if it is polar or non-polar. 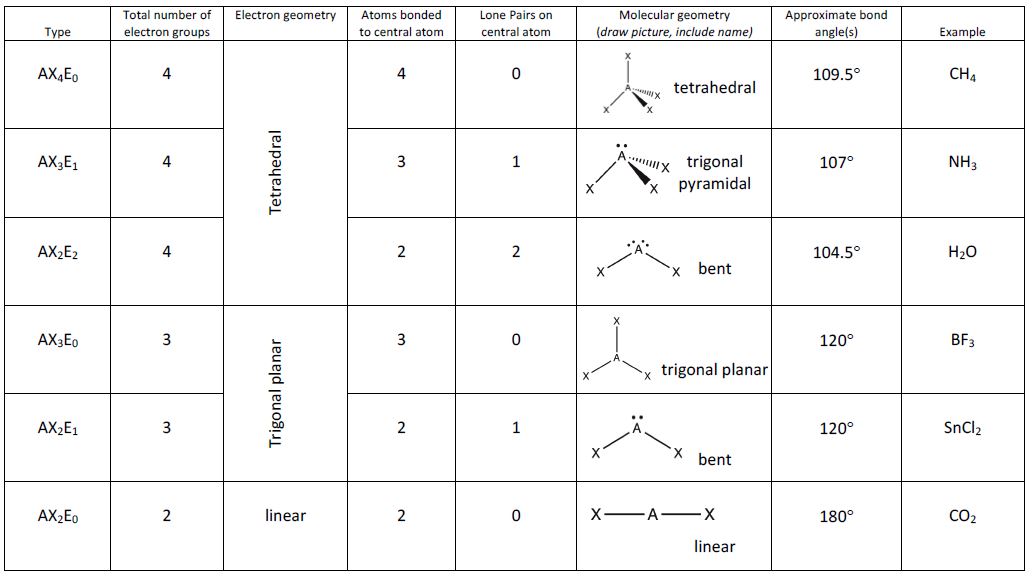
Electronegative ligands such as F will always go to the axial sites.How to Tell if a Molecule is Polar or Non-Polar VSEPR How To Tell if a Molecule Is Polar or Non-Polar? In general, by this reasoning, lone pairs and electropositive ligands such as CH 3 will always prefer the equatorial sites in the trigonal bipyramidal geometry. VSEPR aka Valence Shell Electron Pair Repulsion theory: states that repulsion of electron pairs (both bonds and lone pairs) in the valence shell will dictate. = 0.867 \:bond (formal \: charge = -0.122)\)īecause fluorine is more electronegative than a lone pair, it prefers the axial site where it will have more negative formal charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
